Hydrogen
A replacement fuel
Background
Hydrogen based energy storage systems (ESS) are promising technologies that are currently the subject of much attention. Crucially hydrogen is an “energy vector”, meaning that it can be used as a portable fuel, and the point of production can be decoupled with the point of use. Hydrogen systems are also unusual when compared to other types of energy storage systems in that they use two different processes for the charging and discharging of the energy storage system. Hydrogen production generally involves an electrolyser unit which separates water into hydrogen and oxygen using electricity. Compressed hydrogen gas is usually stored in high pressures tanks, although there are other alternatives like liquefied hydrogen or metal hydrides. Normally an electrochemical device called a Fuel Cell is used to produce electricity, although hydrogen can also be burnt in an internal combustion engine (however there is a large efficiency penalty).
There are many types of fuel cells, such as: Alkaline Fuel Cell (AFC), Polymer Exchange Membrane Fuel Cell (PEMFC), Direct Methanol Fuel Cell (DMFC), Phosphoric Acid Fuel Cell (PAFC), Molten Carbonate Fuel Cell (MCFC), Solid Oxide Fuel Cell (SOFC). The basic differences between these types of batteries are the electrolyte used, their operating temperature, their design, their specific fuel requirement and their field of application.
The Fuel Cell (FC) is the key for this technology. Through an electrochemical process, the fuel is combined with oxygen from the ambient air to produce electricity with heat and water as the waste products. Unlike batteries, FCs will continue to generate electricity as long as a source of fuel is supplied. FCs do not burn fuel, making the electricity generation process quiet, pollution-free and two to three times more efficient than combustion. A single fuel cell consists of two catalyst-coated electrodes (a porous anode and cathode) and an electrolyte in between, similar to a battery. The material used for the electrolyte and the design of the supporting structure determine the type and performance of the FC. The hydrogen used to power them may come from a variety of sources. All FCs work in a similar way.
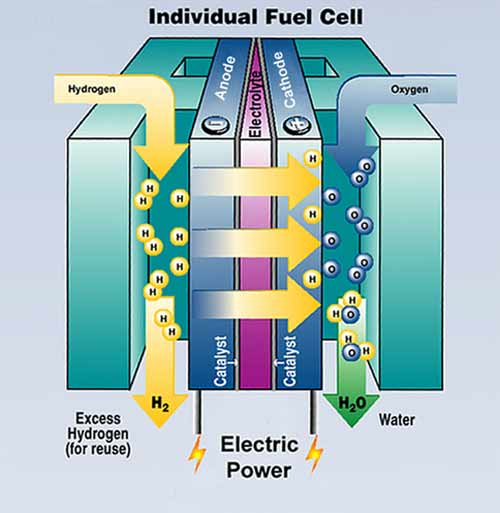
Figure: Individual fuel cell [source US D.O.E., office of energy efficiency and renewable energy]
Characteristics and applications
Hydrogen and fuel cells find applications in longer duration energy storage. This is because their conversion efficiency is very low but they have very negligible self-discharge and hydrogen can hence be stored for a long period of time (though as hydrogen is such a small molecule containment without leakage can be an issue). The high energy density and portability of hydrogen as a fuel also makes it an attractive prospect for (zero emissions) vehicle propulsion in which the waste product is simply water and heat. It is also well suited to peak shaving and energy management applications in which hydrogen is generated at off-peak times or when output from renewable sources is large. This can then be stored for hours or days or months without significant losses and used when the demand is higher. Stationary hydrogen systems are modular and can usually easily allow for further construction/reconfiguration.
Stationary power is the most mature application for FCs. Approximately, 600 systems that produce 10 kW or more have been built and operated worldwide to date. In applications with renewable energy, fuel cells are playing an increasing role. The first and largest plant that integrates hydrogen and wind power has been installed by Norsk Hydro and Enercon in Utsira, Norway, in 2004, which operates as an isolated power system. The system is enough to power 10 houses for 2–3 days without wind. In the town of Nakskov, Denmark, a wind-hydrogen project has been successfully producing hydrogen since May 2007. In the UK the Hydrogen Office in Fife uses a 750 kW wind turbine and a 30 kW Hydrogen electrolyser.
However, despite the fact that hydrogen storage and fuel cells have received large amounts of funding and have been the subject of much research around the world, this technology is still firmly under development and only pilot/demonstration projects have been undertaken. Single fuel cells typically produce low voltages and must be used in series in order to generate large voltages. The fundamental hurdle at present is the very high costs and before large scale deployment can occur, a large reduction is necessary. Recently prices have started to drop and there is a general feeling that manufacturing increases driven especially by applications in the transport/automotive market will reduce the price of fuel cells further. Hydrogen storage also needs further development to allow large quantities of hydrogen to be stored in a manageable volume and thus easily transported. An increase in the durability of fuel cells would also be beneficial, as well as an increase in their tolerance of impurities in the air.
It must also be mentioned that Hydrogen can also be used to supplement natural gas supplies, and burned in the same fashion as natural gas. These systems use excess wind energy to generate hydrogen gas adding it to the natural gas transmission network (there are limits on the amount of hydrogen per unit volume that gas in the transmission network can contain and this has to be monitored). This is a way to “store” excess renewable energy as gas which is then burned and used for heat and cooking etc. Power company E.ON has recently opened a 1 MW prototype system in Hamburg, Germany.
Table of characteristics
| Typical Capacity | Typical Power | Efficiency (%) | Storage Duration | $/kWh | $/kW | Lifespan | Cycling capacity |
| Potential for 100’s MWh | 0-50 MW | 35-45 [3], 20-85 [2] | hours - months | 500 – 1000 [1]6-725 [2], 3-20 [3] | 200 - 400 [1]1500-10000+ [2], 400-1500 [3] | 5-20 years | High |
Table: Hydrogen and fuel cell characteristics